An Aortic-Driven Journey
Our decades of expertise in treating aortic diseases—coupled with our recent acquisitions and partnerships—have empowered us to offer cardiac and vascular surgeons a suite of aortic-centric solutions worthy of their commitment and experience in treating and impacting the lives of patients with diseases of the aorta.
Leading the Advancement
of Treating Aortic Diseases
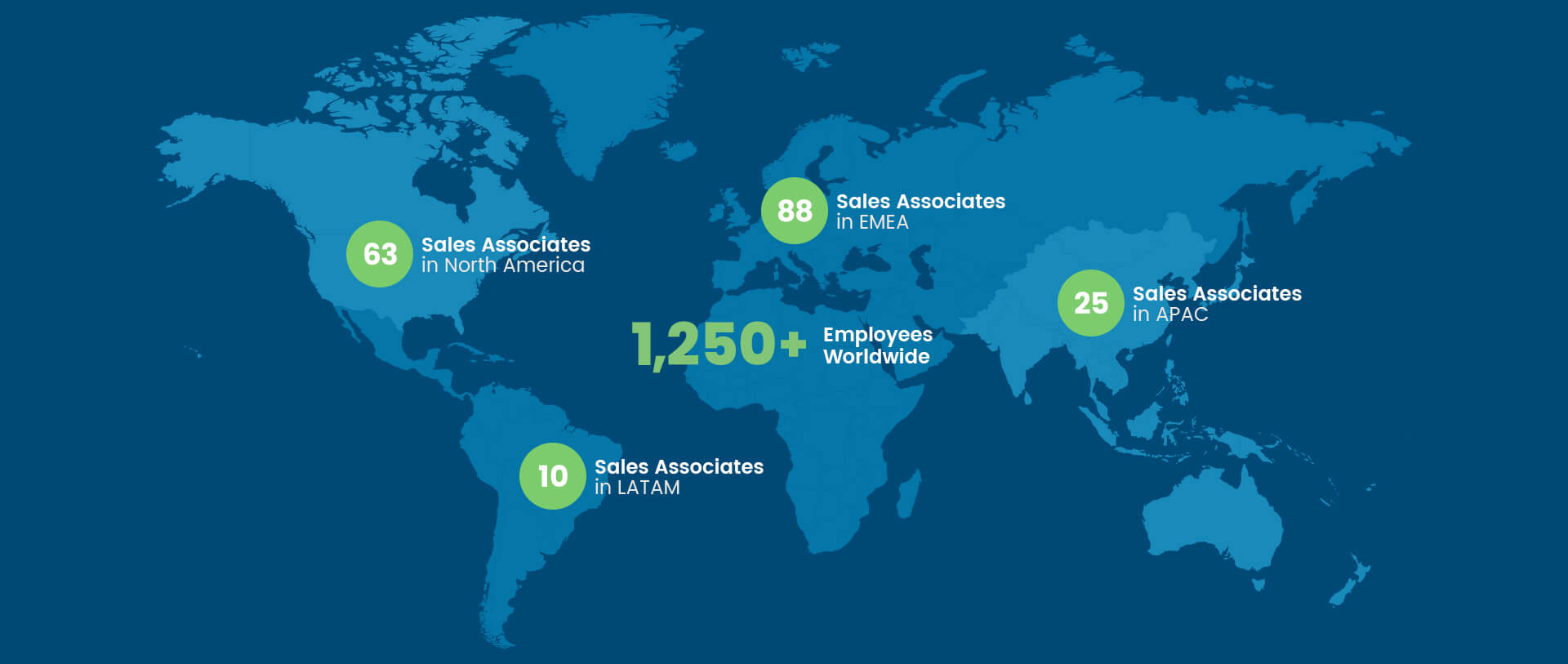
Every day, our 1,300+ global employees dedicate themselves to delivering on the Artivion mission, vision, and core values—working together to form the foundation of an innovative, productive, and sustainable culture.
Our Mission
We partner with surgeons to restore the health of patients by delivering innovative technologies of unsurpassed quality.
Our Vision
To be recognized as a leader in providing technologies for patients with aortic disease.
We are guided by these three key principles and fundamental beliefs:
Collaboration
Working together to deliver for our customers, shareholders, and each other. As we work collaboratively, we’re always truthful and transparent and stand up for what’s right.
Results Driven
Getting it done. Being accountable. Focusing on solutions. Leaning in and bringing out the best in others and always executing with integrity.
Customer Focused
Serving all our customers, externally and internally, with exceptional performance. We dedicate ourselves to treating everyone with respect.
Leadership Team
With decades of combined experience in the medical device industry, Artivion’s leadership team is committed to partnering with cardiac and vascular surgeons to bring innovative aortic technologies of unsurpassed quality to the world, and achieve sustainable revenue growth. In the following profiles, get to know our Executive Team and Board of Directors.

J. Patrick Mackin
Chairman, President, and Chief Executive Officer

Lance Berry
Executive Vice President and Chief Financial Officer

John E. Davis
Chief Commercial Officer

Jean F. Holloway, Esq.
Senior Vice President, General Counsel, Chief Compliance Officer and Corporate Secretary

Marshall S. Stanton, M.D.
Senior Vice President, Clinical Research and Chief Medical Officer

Matthew A. Getz
Vice President, Chief Human Resources Officer

Andrew M. Green
Vice President, Regulatory Affairs

Amy D. Horton, CPA
Vice President, Chief Accounting Officer

Rochelle L. Maney
Vice President, Global Quality

Robert C. Thomson
Vice President, Research and Development

Florian Tyrs
Vice President, Global Operations

J. Patrick Mackin

Thomas F. Ackerman

Daniel J. Bevevino

Marna P. Borgstrom

Jim Bullock

Jeff Burbank

Elizabeth A. Hoff

Jon W. Salveson

Anthony Semedo
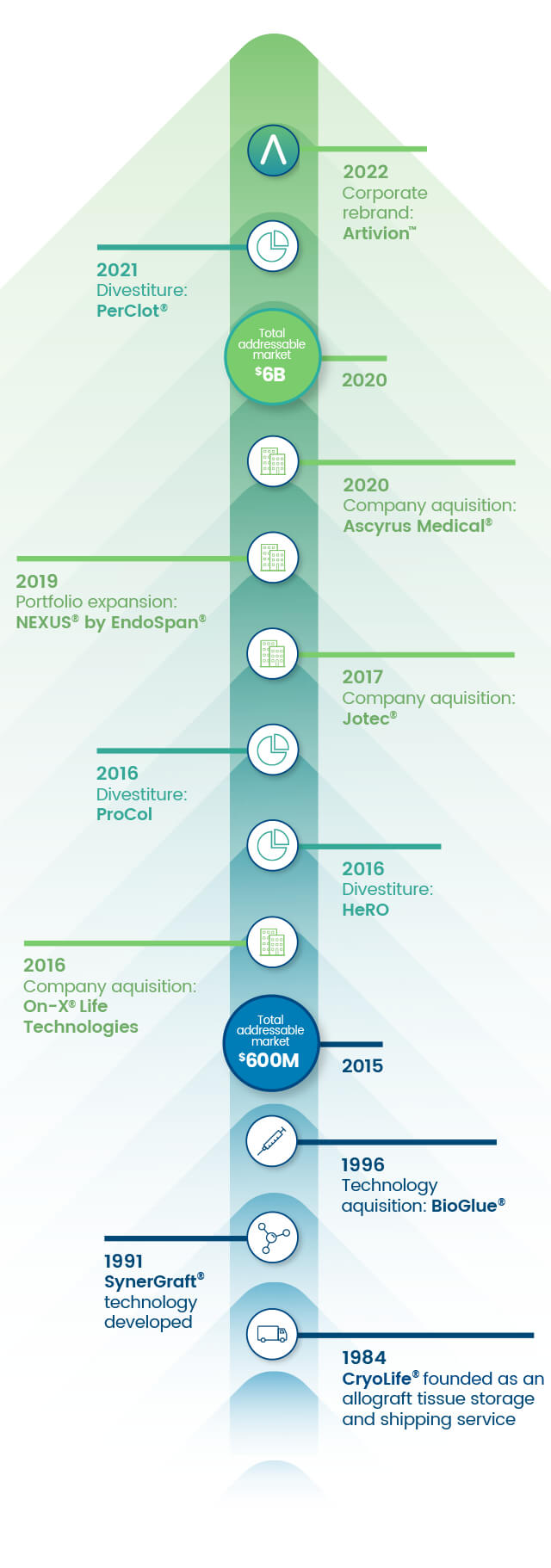
Advancing with Purpose
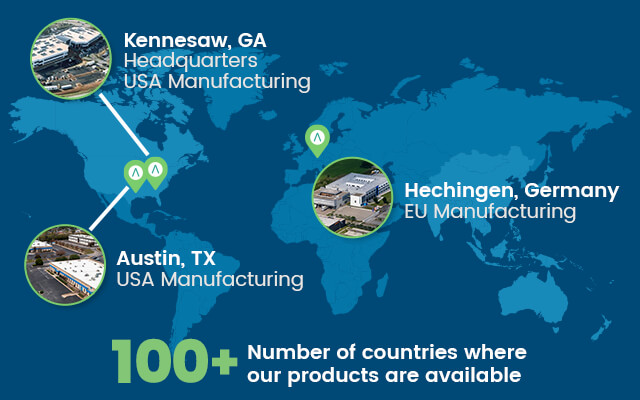
Founded in 1984, we’ve grown into a multi-national organization of over 1,250 employees with sales representation and product manufacturing across the globe.


Outreach Programs
We believe in supporting our clinicians through product donations and fundraising, so they can give back to the global community by operating on patients with limited access to treatment for aortic diseases.
Partnered with 30+ different charitable organizations for 70+ different philanthropic events globally
Supported medical mission trips to 25+ different countries
Donated 500+ cardiac allografts, On-X valves, and other cardiovascular devices
Raised and donated over $70,000 each to Aswan Heart Center in Egypt and Boston Children’s medical mission trip to Ghana
Outreach Programs are conducted in accordance with applicable local and international law and regulation and Artivion’s Code of Ethics and other applicable Company policies. Outreach Programs are conducted without regard to past or potential future sales.